Half Cell vs. Full Cell
What is the difference between a half cell and a full cell in battery science?
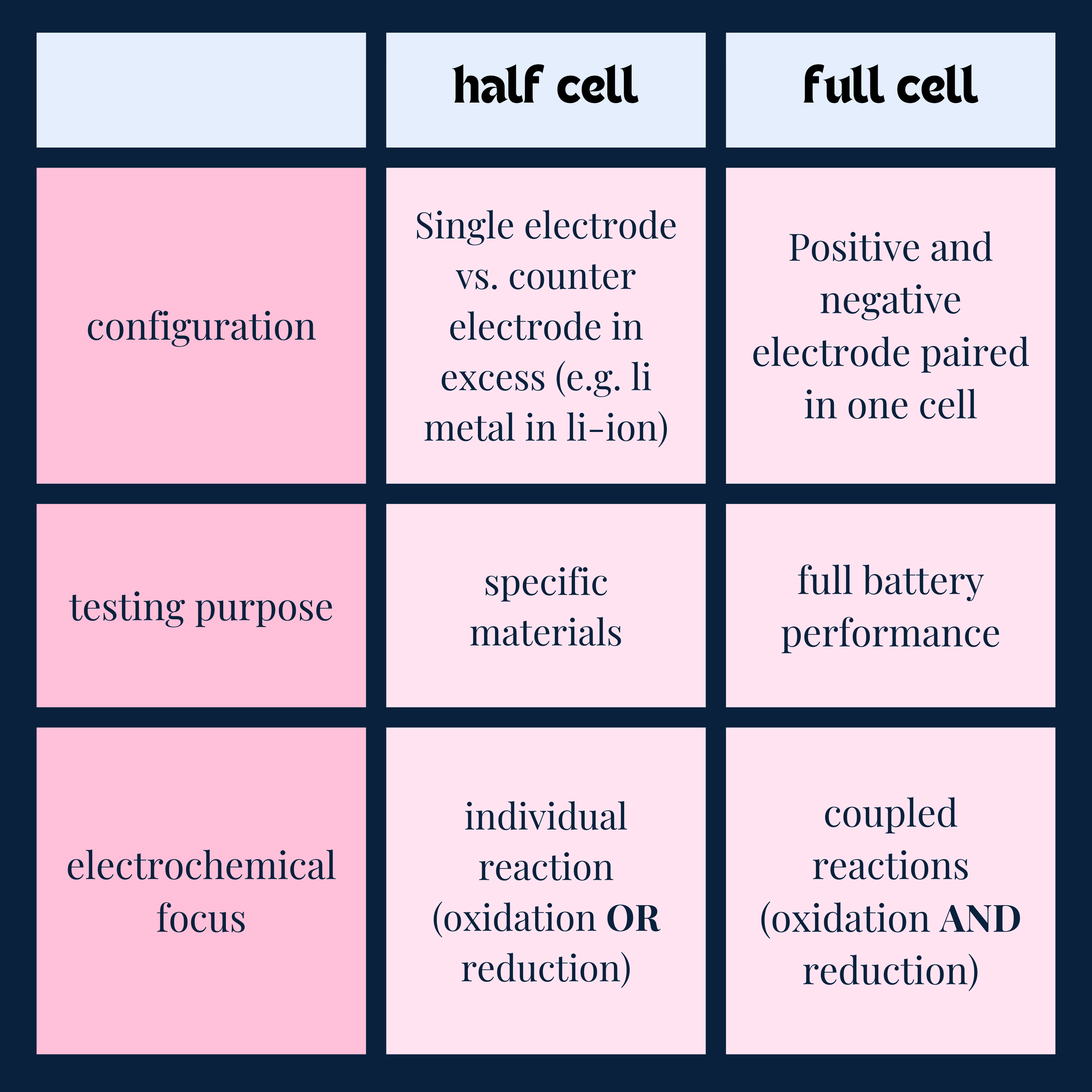
TLDR: a half cell is testing only one commercially viable electrode, and a full cell pairs the two commercial electrodes together to test the complete battery system.
This gets confusing when switching between a Li-ion system and a lithium metal system. When we study solid electrolytes we typically use full cell to mean with the ideal Li metal anode. However in lithium ion cells, a li metal electrode is a half cell, and the li metal is in excess. 🤯🤯🤯
In battery testing, the terms half-cell and full cell refer to different configurations used to evaluate the electrochemical properties of the battery’s components or the complete system.
What is a Half Cell?
A half-cell consists of a single electrode (anode or cathode) paired with a stable reference electrode, such as lithium metal, and an electrolyte.
Purpose: used to test the individual performance of an anode or cathode material in isolation.
Counter Electrode: The working electrode is paired with a counter electrode that is not commercially viable due to it having excess capacity (therefore lowering energy density). In the lithium ion system, lithium metal serves as the counter/reference electrode as it provides the working ion (Li+) in excess.
Measurements: Provides insights into specific capacity, voltage profile, cycle life, and kinetics of the tested material.
Focus: Helps evaluate and optimize electrode materials before integrating them into a full cell.
Li-ion Example: Testing a graphite anode against a lithium metal counter electrode to measure its reversible capacity.
Half Cell vs Full Cell
TLDR: a half cell is testing only one commercially viable electrode, and a full cell pairs the two commercial electrodes together to test the complete battery system.
What is a full cell?
A full cell combines a specific anode and cathode material with an electrolyte to form a complete working battery.
Purpose: evaluates the overall performance of the battery, including energy density, power density, and long-term cycling stability.
Electrodes: Both the anode and cathode materials are application-specific and must balance in capacity for optimal performance.
Measurements: Focuses on the practical operation of the cell, including total capacity, efficiency, and real-world behavior.
Focus: Determines the feasibility of pairing the tested materials for commercial or practical applications.
Li-ion Example: Pairing a graphite anode with an NMC cathode to test the complete battery performance.
In summary, half-cells are ideal for research and development of electrode materials, while full cells are used to test the overall performance and compatibility of the battery system.